|
HELLP syndrome is a potentially life-threatening disorder in pregnancy that is usually associated with preeclampsia, a condition that occurs in 5-8 percent of. This is the most complete list of substances and how they will/may show up in drug screens.to include. Substances that may cause a false positive. What Can Cause A False Positive Drug Test? Search what foods, medications and prescriptions causes a false positive drug test. Learn more about uses, side effects, interactions and warnings for the most common medicines in the UK. Diazepam is a type of medicine called a benzodiazepine. Benzodiazepines are used for their sedative, anxiety-relieving and muscle-relaxing effects. Midazolam 5mg/ml, solution for injection - Summary of Product Characteristics (SPC)Midazolam 5 mg/ml, solution for injection. Midazolam 5 mg/ml contains. Midazolam hydrochloride 5. Midazolam. Titration is strongly recommended to safely obtain the desired level of sedation according to the clinical need, physical status, age and concomitant medication. In adults over 6. Standard dosages are provided in the table below. Additional details are provided in the text following the table. 
Indication. Adults < 6. Adults . The onset of sedation may vary individually depending on the physical status of the patient and the detailed circumstances of dosing (e. If necessary, subsequent doses may be administered according to the individual need. The onset of action is about 2 minutes after the injection. Maximum effect is obtained in about 5 to 1. Adults The IV injection of midazolam should be given slowly at a rate of approximately 1 mg in 3. In adults below the age of 6. Further doses of 1 mg may be given as necessary. Mean total doses have been found to range from 3. A total dose greater than 5 mg is usually not necessary. In adults over 6. Further doses of 0. Since in these patients the peak effect may be reached less rapidly, additional midazolam should be titrated very slowly and carefully. A total dose greater than 3. Paediatric population IV administration: midazolam should be titrated slowly to the desired clinical effect. The initial dose of midazolam should be administered over 2 to 3 minutes. One must wait an additional 2 to 5 minutes to fully evaluate the sedative effect before initiating a procedure or repeating a dose. If further sedation is necessary, continue to titrate with small increments until the appropriate level of sedation is achieved. Infants and young children less than 5 years of age may require substantially higher doses (mg/kg) than older children and adolescents. For this reason, the use in conscious sedation in children less than 6 months of age is not recommended. A total dose up to 0. Prolonged sedation and risk of hypoventilation may be associated with the higher doses. A total dose of up to 0. Prolonged sedation and risk of hypoventilation may be associated with the higher doses. Rectal administration: the total dose of midazolam usually ranges from 0. Rectal administration of the ampoule/vial solution is performed by means of a plastic applicator fixed on the end of the syringe. If the volume to be administered is too small, water may be added up to a total volume of 1. Total dose should be administered at once and repeated rectal administration avoided. The use in children less than 6 months of age is not recommended, as available data in this population are limited. IM administration: the doses used range between 0. A total dose greater than 1. This route should only be used in exceptional cases. Rectal administration should be preferred as IM injection is painful. In children less than 1. Higher concentrations should be diluted to 1 mg/ml. ANAESTHESIA DOSAGE PREMEDICATION Premedication with midazolam given shortly before a procedure produces sedation (induction of sleepiness or drowsiness and relief of apprehension) and preoperative impairment of memory. Midazolam can also be administered in combination with anticholinergics. For this indication midazolam should be administered IV or IM, deep into a large muscle mass 2. Close and continuous monitoring of the patients after administration of premedication is mandatory as interindividual sensitivity varies and symptoms of overdose may occur. Adults For preoperative sedation and to impair memory of preoperative events, the recommended dose for adults of ASA Physical Status I & II and below 6. IV repeated as needed, or 0. IM The dose must be reduced and individualised when midazolam is administered to adults over 6. The recommended initial IV dose is 0. 
A dose of 0. 0. 25 to 0. IM is recommended. In case of concomitant administration of narcotics the midazolam dose should be reduced. The usual dose is 2 to 3 mg. Paediatric population. Neonates and children up to 6 months of age: The use in children less than 6 months of age is not recommended as available data are limited. Children over 6 months of age. Rectal administration: The total dose of midazolam, usually ranging from 0. Rectal administration of the ampoule solution is performed by means of a plastic applicator fixed on the end of the syringe. If the volume to be administered is too small, water may be added up to a total volume of 1. IM administration: As IM injection is painful, this route should only be used in exceptional cases. Rectal administration should be preferred. However, a dose range from 0. IM has been shown to be effective and safe. In children between ages 1 and 1. In children less than 1. Higher concentrations should be diluted to 1 mg/ml. INDUCTION Adults If midazolam is used for induction of anaesthesia before other anaesthetic agents have been administered, the individual response is variable. The dose should be titrated to the desired effect according to the patient's age and clinical status. When midazolam is used before or in combination with other IV or inhalation agents for induction of anaesthesia, the initial dose of each agent should be significantly reduced, at times to as low as 2. The desired level of anaesthesia is reached by stepwise titration. The IV induction dose of midazolam should be given slowly in increments. Each increment of not more than 5 mg should be injected over 2. Midazolam 5mg/ml, solution for injection - Summary of Product Characteristics (SPC) by hameln pharmaceuticals ltd.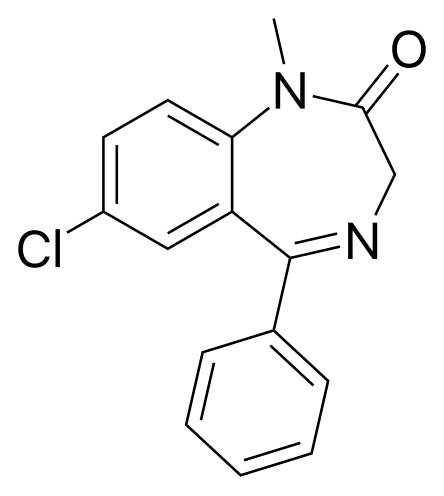
List of drugs that cause false positives on drug tests with reference sources and feedback. Peter Lehmann's List of psychiatric drugs with active ingredients and world-wide trade names. Find patient medical information for Solu-Cortef Injection on WebMD including its uses, side effects and safety, interactions, pictures, warnings and user ratings. In non- premedicated adults below the age of 6. IV). If needed to complete induction, increments of approximately 2. Induction may instead be completed with inhalational anaesthetics. In resistant cases, a total dose of up to 0. Non- premedicated adults over 6. Non- premedicated patients with severe systemic disease or other debilitation usually require less midazolam for induction. An initial dose of 0.  
SEDATIVE COMPONENT IN COMBINED ANAESTHESIA Adults Midazolam can be given as a sedative component in combined anaesthesia by either further intermittent small IV doses (range between 0. IV midazolam (range between 0. The dose and the intervals between doses vary according to the patient's individual reaction. In adults over 6. SEDATION IN INTENSIVE CARE UNITS The desired level of sedation is reached by stepwise titration of midazolam followed by either continuous infusion or intermittent bolus, according to the clinical need, physical status, age and concomitant medication (see section 4. Adults IV loading dose: 0. Each increment of 1 to 2. In hypovolaemic, vasoconstricted, or hypothermic patients the loading dose should be reduced or omitted. When midazolam is given with potent analgesics, the latter should be administered first so that the sedative effects of midazolam can be safely titrated on top of any sedation caused by the analgesic. IV maintenance dose: doses can range from 0. In hypovolaemic, vasoconstricted, or hypothermic patients the maintenance dose should be reduced. The level of sedation should be assessed regularly. With long- term sedation, tolerance may develop and the dose may have to be increased. Paediatric population. Neonates and children up to 6 months of age: Midazolam should be given as a continuous IV infusion, starting at 0. The rate of infusion should be carefully and frequently reassessed, particularly after the first 2. Careful monitoring of respiratory rate and oxygen saturation is required. Children over 6 months of age In intubated and ventilated paediatric patients, a loading dose of 0. IV should be administered slowly over at least 2 to 3 minutes to establish the desired clinical effect. Midazolam should not be administered as a rapid intravenous dose. The loading dose is followed by a continuous IV infusion at 0. The rate of infusion can be increased or decreased (generally by 2. IV doses of midazolam can be administered to increase or maintain the desired effect. When initiating an infusion with midazolam in haemodynamically compromised patients, the usual loading dose should be titrated in small increments and the patient monitored for haemodynamic instability, e. These patients are also vulnerable to the respiratory depressant effects of midazolam and require careful monitoring of respiratory rate and oxygen saturation. In premature infants, neonates and children less than 1. Higher concentrations should be diluted to 1 mg/ml. Special populations. Renal Impairment. In patients with renal impairment (creatinine clearance < 1. IV dose is similar to that reported in healthy volunteers. However, after prolonged infusion in intensive care unit (ICU) patients, the mean duration of the sedative effect in the renal failure population (shown after prolonged infusion in intensive care unit (ICU) patients) was considerably increased most likely due to accumulation of . Therefore the clinical effects may be stronger and prolonged. The required dose of midazolam may be reduced and proper monitoring of vital signs should be established. Use of this drug for conscious sedation in patients with severe respiratory failure or acute respiratory depression. Midazolam should be administered only by experienced physicians in a setting fully equipped for the monitoring and support of respiratory and cardiovascular function and by persons specifically trained in the recognition and management of expected adverse events including respiratory and cardiac resuscitation. Severe cardiorespiratory adverse events have been reported. These have included respiratory depression, apnoea, respiratory arrest and/or cardiac arrest. Such life- threatening incidents are more likely to occur when the injection is given too rapidly or when a high dosage is administered (see section 4. Special caution is required for the indication of conscious sedation in patients with impaired respiratory function. When midazolam is used for premedication, adequate observation of the patient after administration is mandatory as interindividual sensitivity varies and symptoms of overdose may occur.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
November 2017
Categories |
 RSS Feed
RSS Feed
